
|
BRHS /
Hydrogen Ignition
IntroductionThere are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical sparks, fused wires, incendiaries, hot surfaces, heating, rapid adiabatic compression, shock waves and catalytic materials. All of these processes heat a portion of the combustible mixture to a sufficiently high temperature such that adjacent un-combusted layers also react, producing a flame, which propagates through out the mixture. Basic combustion properties
Technical report ISO/TR 15916 (2004) presents basic combustion properties of hydrogen in air mixtures. These are reproduced in the Fig 1-16 below The flammability of hydrogen in air with nitrogen as a diluent is presented by Molnárné et al. (2003), and flammability diagrams are presented at temperatures from 20 °C to 400 °C, and pressures from atmospheric to 100 bar. The flammability diagram for 20 °C and atmospheric pressure is given in Fig 1-20 below, reproduced from Molnárné et al. (2003), with permission. The limiting oxygen concentration at 20 °C and atmospheric pressure can be read from Fig 1-20 as 20.4 % air. Since air contains 21 % oxygen, the limiting oxygen concentration is 0.204 x 21 % = 4.3 % oxygen. Raising the temperature widens the limits, but the lower limit has the greater ratio of change. The major change is to increase the upper limit, and decrease the limiting oxygen concentration (LOC). The diagram at 400 °C and atmospheric pressure is as in Fig 1-21. Note that the limiting oxygen concentration has fallen to 6.2 % air, corresponding to a LOC of 1.3 %, and the upper limit has risen from 75.6 % to 87.6 %. The lower explosive limit has fallen from 4.1 to 1.4 %. Similar effects are shown for increases in pressure, with the limits generally falling with pressure, and the limiting oxygen concentration rising. Tabulated data for the effect of temperature at atmospheric pressure is shown below in Fig 1-17, and the effect of pressure at 20 °C is shown below in Fig 1-18 and Fig 1-19.
Fig 1-17 - Effect of Temperature on Hydrogen Flammability - DIN 51649-1
Fig 1-18 - Effect of Pressure on Hydrogen Flammability - 2.7 dm3 Bomb
Fig 1-19 - Effect of Pressure on Hydrogen Flammability - prEN 1839-B Note that there are small discrepancies between the Tables for 20 °C and 1 bar. This is because the limit determinations were carried out using different methods. The preferred method is that of EN 1839-B.  Fig 1-20 - Flammability Diagram for Hydrogen/Air/Nitrogen at 20 °C and 1 bar  Fig 1-21 - Flammability Diagram for Hydrogen/Air/Nitrogen at 400 °C and 1 bar For hydrogen, the minimum ignition energy is low at 0.017 mJ for mixtures with air (ISO, 2004), and even lower at 0.0012 mJ for mixtures with oxygen (Kuchta, 1986). Hydrogen has a such a low minimum ignition energy that it is often difficult to determine the exact mechanism and cause of an ignition when it occurs. In some incidents where hydrogen has been involved in an explosion or fire, it has not been certain as to the source of ignition and the mechanism of the release. Some incidents have had obvious ignition sources such as flames or grinding sparks, but other incidents had releases of hydrogen where all obvious sources of ignition had been excluded, and self ignition had been blamed. The propensity of hydrogen to ignite in this fashion for no apparent reason has been reported several times previously, by Reider (1965), Lees, (1991), Anon (1922), and Fenning and Cotton (1930). In these incidents, no specific cause for ignition was identified. Several mechanisms have been suggested, of which two are worthy of note for discussion. The first mechanism is that of the reverse Joule-Thomson effect exhibited by hydrogen, and the second, the so-called "diffusion ignition" mechanism.
Static electricityThe ignition hazards posed by static electricity require specific precautions to taken. A Technical Report by CENELEC (2003) does not specifically refer to hydrogen but does refer to the flammable gas Group IIC and the ignition hazards associated with static electricity. This report addresses the maximum areas of insulating materials that may become charged, and restricting the maximum area limits the maximum charge that can be transferred from the surface in the form of a brush discharge. Such a discharge does not have a specific energy, but the ignition properties can be compared to those of a spark discharge between two conductors. The comparison has been discussed by Gibson and Harper (1988), who proposed the term incendivity to compare the quantity of charge transferred with its capability to ignite a flammable mixture with a given minimum ignition energy. Further work by von Pidoll et al. (2004) gives specific guidance on the maximum tolerable charge transfer to ensure freedom from ignition. However, he has correlated the charge transferred with minimum ignition energy and the gas groups defined by the IEC in their standard, but does not take account of the time and spatial characteristics of the discharge. The maximum tolerable charge transferred for hydrogen is given by the CENELEC (2003) as 10 nC. This is a very small quantity of charge, and hence hydrogen is extremely prone to ignition from electrostatic discharges from insulating or non-conductive materials. When using non-conductive solid materials there are restrictions on the size of chargeable surfaces. The restriction on the size of chargeable surfaces depends on the ignitability of the gases and vapours and the classification of the hazardous area:
The restrictions on areas are given in the following tables:
Fig 1-22 - Restriction on chargeable surface depending on zones and gas categories
Fig 1-23 - Restrictions on widths of narrow materials depending on zones and gas categories There have been several instances in the past where accidental hydrogen releases have ignited spontaneously. Whilst these have been investigated, no satisfactory explanation has been produced, but there have been suggestions that some form of electrostatic charging has been present, resulting in an ignition. In view of the very low ignition energy of hydrogen, such ignitions are a distinct possibility. Astbury and Hawksworth (2005) have undertaken a critical review of several incidents with their postulated mechanisms, and has concluded that there is a distinct possibility that releases which ignite spontaneously may be of an electrostatic origin. Whilst electrostatics can be due to an obvious charging mechanism, such as the discharge from a sheet of insulating plastic as described above, ignition of hydrogen from electrostatics generated from or by the hydrogen itself is a far more difficult mechanism to define. Generally, there is no particular propensity for pure gases to become electrostatically charged, as described by CENELEC (2003). As reported by Astbury and Hawksworth (2005), work at the U.K. Health and Safety Laboratory on releasing high pressure (15 MN m2) hydrogen through various nozzles from 0.5 to 12 mm in diameter did not result in any spontaneous ignition at all. This would suggest that straightforward pure hydrogen does not in itself ignite. However, there have been reports, particularly one by the periodical Engineering (Anon, 1930) reporting work undertaken by Nusselt in Germany, after a spontaneous ignition of hydrogen. Much of this work centred on the possibility of a catalytic effect from fine rust that was present. However, when hydrogen was released in the dark through a nozzle fitted with a funnel and a wire probe, corona discharges were seen, and on tapping the nozzle to disturb the rust, an ignition took place. Clearly under these circumstances the corona discharge was sufficient to ignite the released hydrogen. It is likely that the corona was started by the movement of the hydrogen generating an electrostatically charged dust cloud which was sufficient to produce an electrostatic field of a strength high enough to initiate a corona from the wire inserted into the funnel.
Electric sparksElectrical sparks are defined as discontinuous electrical discharges across a gap, in an otherwise complete electric circuit, between at least two electrodes occurring when the voltage exceeds the breakdown voltage. The characteristics of the discharge depend very much on the discharge circuit involving capacitors, resistors and inductors. In many technical applications, especially in spark ignition engines, optimized, standard ignition systems are used. Typically the systems are based on a capacitor to store the energy supplied by a high voltage generator, complemented by a circuit of low induction and resistance containing an electronic device triggering the spark for control and synchronization purposes. In Fig 1-24 a schematic diagram of the voltage and current of ignition spark as a function of time is presented after Maly and Vogel (1978). 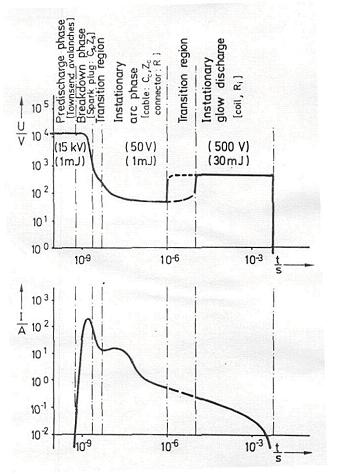 Fig 1-24 - Schematic diagrams of voltage and current of technical ignition systems as functions of discharge time. Typical values are given in parentheses, circuit parameters responsible for a discharge mode are indicated in brackets As it can be seen a typical electrical spark of a commercial ignition system can be divided in three phases. First a breakdown phase is initiated which creates in the gas a small diameter (~40 mm) conducting plasma channel between the two electrodes. The phase is very short (1-10 ns) and characterized by high voltages (~10 kV) and currents (~200 A). Temperatures in the plasma channel reach up to 60 000 K, the molecules are fully dissociated and ionized causing the pressure to jump to 200 bar creating an intense shock wave and expansion of the discharge zone. The high conductivity of the plasma reduces the voltage provided by the circuit and the arc phase is initiated. The arc voltage is low (<100 V) although the current may be as high as the electrical circuit permits (up to several kA). The maximum temperature in the discharge zone drops to about 6000 K and ionization falls to a 1 % level. The arc expands mainly due to conduction and diffusion producing bell shaped temperature and ionization profiles. Heat losses create appreciable cathode and anode voltage falls and the glow phase begins during which currents drop to a 200 mA level, the gas temperature is less than 3000 K and ionization less than 0.01 %. The conversion efficiency of electrical to thermal energy of the gas decreases from the breakdown to the glow phase mostly due to the heat losses to the electrodes. To initiate a flame shortly after breakdown the chemical reaction must produce enough energy to overcome heat losses and the ignition kernel has to grow beyond a critical size for the flame to develop, typically must be two times larger than the laminar flame thickness. Obviously the electrodes, their separation, shapes and material play an important role in this process. 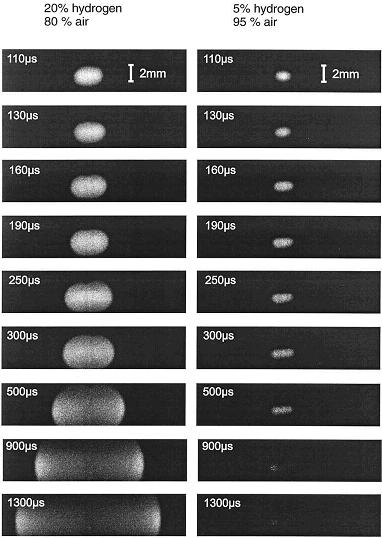 Fig 1-25 - Temporal evolution of a flame kernel visible as a relative OH concentration profiles recorded by PLIF for an ignitable and below the ignition limit mixture of hydrogen and air. Spark duration was 100 ms Spark ignition constitutes a very complex interplay between plasma kinetics, chemical kinetics, molecular transport processes and fluid dynamics. Experimental investigations of spark kernels and their transition to flame kernels are rendered difficult because of very short process times, extremely high core temperatures and large gradients in the refractive index. In general, only flame propagation subsequent to spark ignition can be studied by laser diagnostic methods. Therefore, internal structures of the plasma core remain mostly unknown. A complete mathematical simulation of a spark ignition is also a difficult task because of the enormous numerical problems, due to the stiffness and high dimensionality of the problem (each chemical species introduces an additional conservation equation). Recently Thiele et al. (2002) have conducted detailed two-dimensional numerical modeling of spark ignition of hydrogen-air mixtures. Their model considered heat conduction from the gas phase to the electrodes, detailed chemistry and molecular transport as well as the coupling of the gas dynamics to the properties of the electrical discharge through heating by the electrical current. They also performed spark ignition experiments using a highly reproducible ignition system. Shapes of early flame kernels were monitored by 2-D laser-induced fluorescence PLIF imaging of OH radicals produced during the ignition and the combustion process (Fig.2). In addition, for a central position within flame kernel, temperatures were measured using Coherent Anti-Stokes Raman spectroscopy (CARS). Results from experiments and simulations suggest the birth of a self sustaining flame propagation for process times between 50 to 70 ms after arc breakdown.
Auto-ignitionGenerally auto-ignition results from either the exothermic of chain branching character of the oxidation reactions that at certain conditions self-accelerate to reach high conversion and heat release rates. Auto-ignition limits can be established testing experimentally or theoretically a homogeneous mixture of volume V filling a vessel whose walls have a temperature Tw. Once the heat release rate in the volume due to reactions exceeds the heat lost to the walls or if the reaction rates in the vessel exceed the reaction quenching (termination) rates by the walls or in the gas a thermal or branched-chain (isothermal) auto-ignition occurs. Typically, as almost all combustion reactions are exothermic, chain auto-ignitions cause also self heating and are accelerated by both factors. Obviously auto-ignition limits are not only a feature of the mixture composition and parameters (pressure, temperature) but also of the vessel size, wall properties and internal flow conditions. 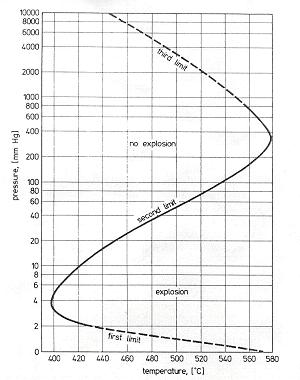 Fig 1-26 - Explosion limits of a stoichiometric hydrogen-oxygen mixture in a spherical HCl coated vessel of 7.4 cm diameter This is illustrated in Fig 1-26 showing the auto-ignition limits often called also explosion limits for a stoichiometric mixture of hydrogen and oxygen (B. Lewis, 1987) providing the important parameters of the test vessel. We may note the logarithmic scale of pressure and linear scale of temperature showing that pressure effects on reaction rates are weaker than temperature effects as one would expect by the consideration of Arrhenius chemistry. The first and second limits, although interesting from the fundamental point of view, correspond to very low pressure (up to about 0.3 bar) and are thus of little practical interest. The third limit follows the trend that one would expect from simple density considerations. As the pressure increases, the initial densities of the reactants increase and a lower temperature is necessary for the reactions to reach a critical reaction rate for explosion. For safety considerations explosions in large volumes where wall effects can be neglected at atmospheric conditions and for most violently reacting, i.e. stoichiometric mixtures are considered. Thus, in typical safety manuals a temperature of 585 °C is given as the auto-ignition temperature for hydrogen air systems. The initial reaction rate in auto-ignition is very small thus a certain time must pass before the reaction has reached a defined rate. This time interval is called ignition delay. Ignition delays are particularly important for operation of engines as they provide the engine speed limits where operation is possible due to auto-ignition (compression ignition engines) or where auto-ignition can be avoided when detrimental (knock in spark ignition engines). Most accurate ignition delay measurements can be performed in shock tubes in wall reflected shocks where the heating of the mixture is practically instantaneous. A research issue is then prediction of the ignition delays using available kinetic data. The state of the art in this field is far from satisfactory as illustrated in Fig 1-27 - after Wang et al. (B. L. Wang, 2003) where a comparison of measured and calculated ignition delay times using different chemical reaction mechanism, available in the literature is provided. 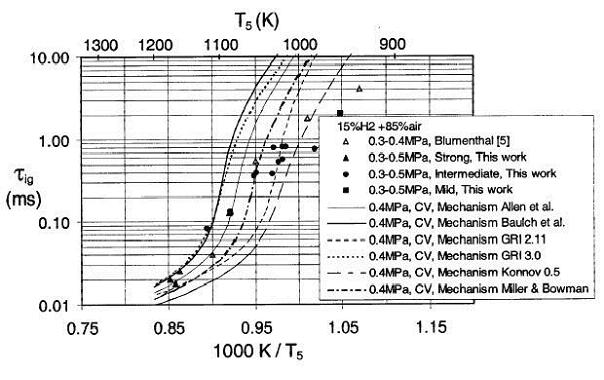 Fig 1-27 - Measured and calculated ignition delay times in a H2-air mixture In the low temperature range the measured ignition delay times are much shorter (even by orders of magnitude) than the theoretically predicted ones. Partially this is because in shock wave ignition mild and strong ignition regimes are possible. For the case of mild ignition multiple and random ignition kernels appear in the mixture. In the case of strong ignition typically reproducible direct transitions to detonation occur. Mild ignition is extremely sensitive to homogeneity of the mixture, impurities, wall properties of the shock tube and particularly to presence of even minute obstacles causing shock reflections, shock focusing and thus generating local hot spots. Thus, this form of ignition is very difficult to control particularly in natural conditions. If the volume of the mixture is large enough deflagration to detonation transition will eventually occur even after mild ignition causing convergence of effects of both types of ignition in accidental explosions. An interesting case of shock wave induced ignition is the jet release auto-ignition that may happen when gas kept at high pressure is rapidly released from a volume into the atmosphere. The flow velocities in the jets reach supersonic levels due to further expansion after the release valve. Then shock waves will appear in the jets due to interaction with the surrounding gas, heating the nascent mixture to temperatures exceeding auto-ignition levels, Wolanski and Wojcicki (1972). If obstacles are in the way of the jets the process is highly facilitated.
Mechanical Friction and ImpactMechanical Rubbing
Brearley and Tolson (1995) measured power levels and contact loads required to ignite flammable gas mixtures by a 25 mm cube of stainless steel frictionally heated through rubbing against a stainless steel wheel at circumferential velocities of 5 and 20 m s-1. In these tests a contact load of 750 N was required to ignite hydrogen. This equates to a dissipated power of approximately 2 kW and a power density of approximately 0.5 W mm-2. No temperature recordings were made in the tests. It was noted that in most cases the ignition was caused by the hot spot close to the point of contact. In Powell’s (1984) review he summarises data from various experiments breaking them into two categories with rubbing speeds above and below 10 m s-1. The Table is presented below and shows results from Group IIC gases (hydrogen, etc). Work at HSL (MECHEX Project) has shown that at low rubbing speeds, hydrogen was ignited at a temperature close to the auto-ignition temperature. The conditions were: power 0.7 kW and rubbing speed 0.7 m s-1 which caused ignition at 530 °C. Ignition occurred from the hot surface with few sparks produced from the low speed conditions. Impact
In his paper, Powell (1986) states how little energy is needed to ignite flammable gases and vapours with impact of light metals and their alloys, producing burning particles with temperatures in excess of 2000 °C from light impacts (<1 J) with material such as Cerium, Titanium, Zirconium, Hafnium and their alloys. Impacts on smears of aluminium or magnesium on rusty steel are also equally incendive. There is therefore a high probability of igniting hydrogen under these conditions. Powell reports on ignitions caused by impacts between very hard steels (>550 VPN). Energies of 250 to 1000 J are required to ignite methane-air, with slightly lower energies igniting IIA vapours. For steel with with VPN of approximately 550, an energy of 180 J was sufficient to cause ignition. Ignition of hydrogen-air mixtures from impacts involving very hard steels are therefore very likely. Mechanical sparks
There are a number of key properties of burning metal particles or sparks that are relevant to their ability to cause ignition of a flammable atmospheres. These include: size, material, velocity, temperature, number, combustion rate and time. There is a metal to metal contact pressure and relative velocity threshold for spark production during impact, rubbing or grinding. Above the threshold metal particles are lost from the weaker of the two materials. Generally, particles are only produced when the relative velocity between the two surfaces exceeds 1 m s-1, Bernend and Ritter (2000).
Ignition by ExplosiveAn explosive is a powerful ignition source and will readily ignite a flammable fuel/air mixture and even under the right conditions directly initiate a detonation in an unconfined mixture. No data has been found in the literature on the minimum amount of explosive required to ignite a fuel/air mixture, but there have been a number of experimental and theoretical studies to determine the minimum amount of explosive required to initiate an unconfined detonation by Bull et al. (1978) and Bull (1979). As might be predicted from the general behaviour with other ignition sources the mass of explosive required to initiate an unconfined detonation in a hydrogen/air mixture is very much less than that required for less reactive fuel/air mixtures such as propane or methane. Table 1 compares the minimum amount of tetryl required to initiate an unconfined detonation in the most detonable hydrogen/air mixture and other common fuel/air mixtures. For hydrogen and the other fuels listed in Table 1, apart from acetylene, the most detonable mixture lies within a stoichiometry range of between 1.1 and 1.3. For acetylene the value for the most detonable mixture is 2.05. For all fuels the minimum amount of explosive required increases rapidly as the mixture approaches the detonation limits. In the case of hydrogen it is predicted there is about a hundred-fold increase in the mass of tetryl required to initiate a detonation in near limit mixtures compared to the most detonable mixture (stoichiometry of about 1.1).
Fig 1-28 - Minimum mass of tetryl required to initiate unconfined detonation
Ignition by Open Flame and Hot SurfaceIgnition by a hot surface occurs as a result of local heating of the hydrogen-oxidant mixture to the point where a sufficently large volume reaches the autoignition temperature and the combustion reaction is initiated. For this to occur generally requires the surface to be at a temperature well above the autoignition temperature, see Powell (1984), although the actual temperature depends on a number of factors in addition to the usual mixture concentration, ambient temperature etc. These additional factors determine the hot surface ignition behaviour of flammable gases and not just hydrogen, and include the size and shape of the hot surface, the degree of confinement around the surface, the strength of the convection currents across the surface, see Laurendau (1982) and the material of the surface, Lewis and von Elbe (1987), page 380). 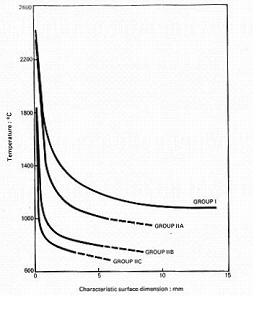 Fig 1-29 - Summarises size dependence of hot surface ignition temperatures as a function of size (Hydrogen is represented by curve IIC) For a particular hot surface, ignition is characterised by an ignition delay, which under ideal circumstances multiplied by the power for ignition gives a linear relationship between the product (energy) and ignition delay (Lewis and von Elbe, 1987, page 365 and Carleton et al. (2000)). The offset on the y-axis in this plot is the minimum power for ignition for that arrangement. The temperatures required to cause ignition of mixtures of hydrogen with air and oxygen, see review in Buckle and Chandra (1996), Carleton et al. (2000) and Hawksworth et al. (2005), range from 640 °C to 930 °C, the spread of temperatures being explained the size, geometry effects etc described in the first paragraph. While the temperatures quoted are above the auto ignition temperature, the increase is much less than seen for hydrocarbon-fuel air mixtures, as illustrated by the IIA curve in Fig 1-29. In terms of simple modelling of hot surface ignition, Laurendau (1982) presents a simple model in terms of a one step reaction chemical kinetics model. Interestingly, the most easily ignited mixture of hydrogen with air lies lean of stoichiometric, see Calcote and Gregory (1952), while work using very small hot surfaces, Carleton et al. (2000) and Hawksworth et al. (2004), suggests that mixtures as low as 10 to 15 % are the most easily ignited. For hydrogen-oxygen mixtures, the work of Buckle and Chandra (1996) indicates a fairly flat H2 concentration dependence (slight positive slope with increasing hydrogen concentration) between roughly 20 and 90 % hydrogen in oxygen. Catalytic surfaces (e.g. plantinium) have a dramatic effect on the ignition temperature required, Cho and Law (1986), with ignitions reported at temperatures as low 70 °C.
MiscellaneousMicrowaves
Lightning
Hydrogen Chemical Incompatibility/Exothermic Reaction
The major hazard with hydrogen (CAS Number 1333-74-0) is its flammability and ease of ignition. In chemical terms, it is relatively stable. However, it is easily oxidised, so it is incompatible with oxidising materials. The most common ones are the halides, particularly fluorine and chlorine, where hydrogen ignites spontaneously if mixed with these materials. In general, any material containing oxygen or other oxidising species may cause a reaction with hydrogen, or catalytic materials such as Raney nickel, activated carbon and precious metal catalysts can cause spontaneous ignition of hydrogen. According to Pohanish and Greene (2003), hydrogen reacts violently, explosively or forms heat and shock sensitive explosive materials when in contact with oxidizers, halogens, acetylene, bromine pentafluoride, chlorine oxides, dinitrogen oxides, fluorine perchloride, heptasilver nitrate octaoxide, iodine heptafluoride, 3-methyl-2-penten-4-yn-l-ol, nitric oxide, nitrous oxide, oxygen difluoride, and other gases. It is incompatible with copper(II) oxide, difluorodiazene, iodine heptafluoride, lead trifluoride, liquid nitrogen, lithium perchlorate trihydrate, metals, nitrogen trifluoride, nitryl fluoride, palladium(II) oxide, palladium trifluoride, polycarbon monofluoride, potassium, tetrafluorohydrazine, xenon hexafluoride, and may cause ignition on contact or explode when heated. The above list is not exhaustive. The other major problem with hydrogen is its ability to embrittled steels, so that the steel cracks. Hydrogen embrittlement is the result of hydrogen that diffuses along the grain boundaries and combines with the carbon to form methane gas. The methane gas collects in small voids along the grain boundaries where it builds up enormous pressures that initiate cracks and decrease the ductility of the steel. If the metal is under stress, brittle fracture can occur. The only method of prevention is to use a suitable steel which has a proven resistance to embrittlement. Further information on hydrogen attack of metals is available from Oriani, Hirth and Smialowski (1985).
Optical radiation
The potential for an explosive atmosphere to be ignited by optical radiation has been extensively investigated during an EU funded project PROPEX (Prenormative Research on the use of Optics in Potentially EXplosive atmospheres). As part of this work a number of potential mechanisms for ignition were identified as listed below (as summarised in draft standard IEC 60079-28/CDV):
The most likely case of ignition occurring in practice with lowest radiation power of ignition capability is case 1. Under some conditions for pulsed radiation case 4 also will become relevant. For the case of hydrogen, scenario a) using Continuous Wave (CW) radiation gave a measured ignition power of 140 mW, using a 1064 nm laser feeding into a 62.5 mm diameter fibre the cleaved end of which was coated with iron oxide. This compared to the most sensitive explosive atmosphere (carbon disulphide) at 50 mW (reduced to 24 mW in later tests) and the least sensitive (n-pentane) at 304 mW. This reference also includes data for larger surface areas which has been used in the draft standard (IEC 60079-28), for Group IIC, T4 and above (includes hydrogen) have a power limit of 35 mW (includes safety factor) and an irradiance of 5 mW mm-2 for surface areas not exceeding 400 mm2. For pulsed radiation, the work of the PROPEX project led to the following guidance in IEC 60079-28:
In terms of other mechanisms listed above, no data is currently available, and so careful consideration is required if for example a UV beam were to be passed through an explosive hydrogen mixture.
Acoustics
There is no data on the ignition of hydrogen due to acoustic effects. Ignition of Liquid Hydrogen and Solid Oxygen MixturesLiquid hydrogen has properties which set it apart from the other cryogenic fuel of liquefied natural gas, in that its atmospheric boiling point is much lower than that of air. Pipe-work and vessels at liquid hydrogen temperatures of about 20 K will condense air from the atmosphere. It will also be cold enough to condense and freeze water and carbon dioxide from the air.
Fig 1-30 Data from Yarwood and Castle (1961), except #National Research Council (1933) Work undertaken by Perlee et al. (1964) indicated that liquid hydrogen can condense and freeze oxygen. The resultant solid oxygen in an excess of liquid hydrogen can be detonated by impact. They used a rifle and with a muzzle velocity of 600 m s-1, detonation always occurred when the bullet impacted the mixture. Detonation was indicated by the use of thin metal strip gauges which deflected permanently when a detonation occurred. The results indicated that the explosive yield of liquid hydrogen-solid oxygen mixtures was greater than those for equal weights of trinitrotoluene (TNT). Various experiments were carried out in which there was a large excess of liquid hydrogen. The specific gravity of liquid hydrogen is typically about 0.07, and that for the solid oxygen in the a state is about 1.426. The sonic velocities in the hydrogen and solid oxygen phases ... [more text to follow]
<< Knowledge Gaps and Recent Progress | Content | Contributing Authors >> |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||